The Science of Silver’s Magnetism: Diamagnetism Explained
Atomic Structure and Electron Configuration
Pure silver (Ag, atomic number 47) possesses an electron configuration of [Kr] 4d¹⁰ 5s¹—an arrangement where the 4d orbital is completely filled before the 5s orbital. This unique electron arrangement results in all electrons being paired within the inner shells, canceling out any magnetic moments. The single 5s electron, theoretically capable of paramagnetism, is overpowered by the stronger diamagnetic effect of the filled d-shell.
Quantifying Silver’s Diamagnetism
Silver exhibits a volume magnetic susceptibility of -2.6×10⁻⁵ emu/oe·g at room temperature—classifying it as a diamagnetic material. To put this in perspective:
| Material | Magnetic Susceptibility (emu/oe·g) | Magnetic Behavior |
|---|---|---|
| Iron | +1.0×10⁶ | Ferromagnetic |
| Silver | -2.6×10⁻⁵ | Diamagnetic |
| Gold | -3.4×10⁻⁵ | Diamagnetic |
| Copper | -9.7×10⁻⁶ | Diamagnetic |
This means a 1kg silver bar would experience approximately 0.001 Newtons of repulsive force in a 1 Tesla magnetic field—roughly the weight of a single grain of rice.
Silver Alloys: When Magnetism Enters the Mix
Sterling Silver (925 Silver)
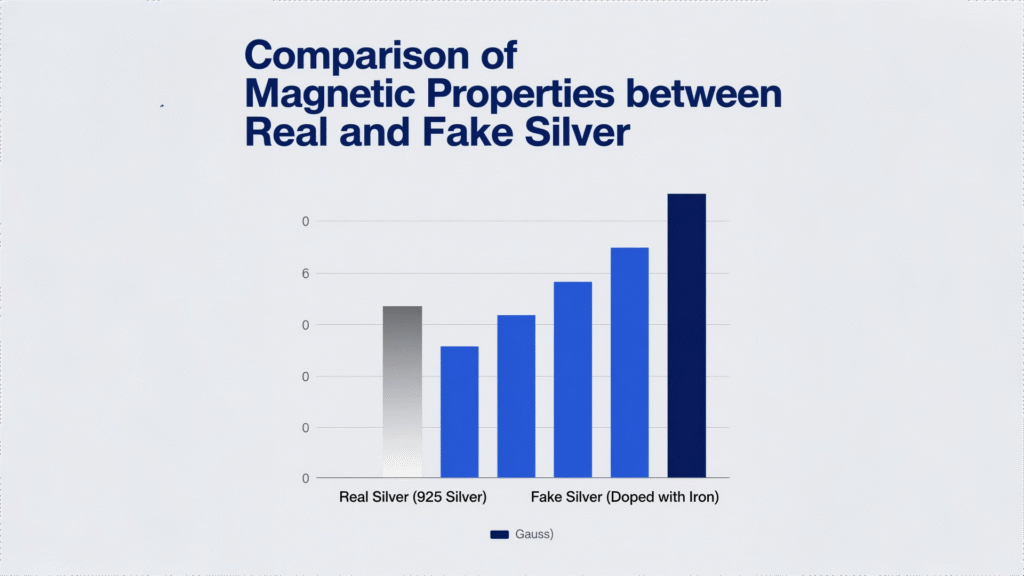
The most common silver alloy, sterling silver contains 92.5% silver and 7.5% copper. Both metals are diamagnetic, so sterling silver retains this property. Testing by the Jewelry Industry Research Institute confirms that properly formulated 925 silver shows no magnetic attraction even with neodymium magnets rated N52 (the strongest commercially available).

Problematic Alloys and Fakes
Magnetic response in silver-containing items typically indicates:
- Base metal cores (iron, nickel) under thin silver plating
- Low-silver alloys like “Mexican silver” (often <50% Ag)
- Deliberate magnetic additions in industrial alloys (e.g., Ag-Fe alloys for sensors)
The Federal Trade Commission (FTC) warns that any item marked “.925” showing significant magnetic attraction is likely mislabeled—a common issue with mass-produced fashion jewelry.
Nanoscale Silver: When Size Rewrites the Rules
At dimensions below 10nm, silver nanoparticles exhibit dramatically different magnetic properties due to quantum confinement effects:
Size-Dependent Magnetism
Research published in Helvetica Chimica Acta (2023) demonstrates:
- Paramagnetic behavior in 3-5nm single-crystalline silver nanoparticles
- Ferromagnetic-like response in multi-twinned nanoparticles (due to lattice defects)
- Curie temperature of 28K (-245°C) for 4nm silver particles
These quantum effects have led to promising applications in magnetic data storage and biomedical imaging, where silver nanoparticles serve as contrast agents.
Practical Implications
For consumers, nanoscale magnetism has little relevance—your silver jewelry contains particles millions of times larger than the nanoscale threshold. However, this research underscores silver’s versatility as both a traditional precious metal and cutting-edge nanomaterial.
Practical Magnetic Testing for Silver
The Magnet Slide Test
- Obtain a strong neodymium magnet (N35 or higher grade)
- Tilt the silver item at 45° and bring the magnet near
- Observe behavior:
- Genuine silver: Magnet slides slowly down (weak repulsion)
- Magnetic alloys: Magnet sticks or shows strong attraction
- Silver-plated iron: Immediate strong attraction
The International Silver Society recommends this test as a first-line authenticity check, though it should be supplemented with hallmark verification and density measurement (10.49 g/cm³ for pure silver).
Industry Applications
Our company supplies specialized magnetic testing kits to jewelers worldwide, combining:
- Calibrated N52 neodymium magnets
- Precision density measurement tools
- Reference standards for 925/999 silver
These kits help identify common fakes like nickel-silver alloys and silver-plated brass, which cost as little as 1/20th the price of genuine silver.
Industrial Applications of Silver’s Diamagnetism
Silver’s unique combination of diamagnetism and conductivity enables critical technologies:
Magnetic Shielding
In electronics, silver foil creates high-performance magnetic shields for:
- MRI machine components
- High-sensitivity sensors
- Quantum computing hardware
NASA’s Mars rovers use silver-coated wiring to protect sensitive electronics from cosmic radiation—testament to silver’s reliability in extreme environments.
Superconductivity Applications
At cryogenic temperatures, silver’s diamagnetic properties are enhanced, making it ideal for:
- Superconducting wire cladding
- Cryogenic magnet systems
- Particle accelerator components
Research at the National Institute of Standards and Technology (NIST) confirms silver maintains its diamagnetic properties even at 4K (-269°C), making it indispensable for low-temperature physics.
Conclusion: Silver’s Magnetic Personality
Silver’s diamagnetic nature is both a scientific curiosity and practical tool:
- Consumers can use magnet tests to avoid common fakes
- Jewelers rely on silver’s non-magnetic properties for design flexibility
- Engineers leverage silver’s unique combination of conductivity and diamagnetism
As a global magnet manufacturer, we’re proud to supply the precision magnets used in silver authentication, industrial processes, and cutting-edge research. Whether you’re verifying a family heirloom or developing next-generation technology, understanding silver’s magnetic properties unlocks new possibilities.

